The Silent Disruption: How Oral GLP-1 Peptide Therapeutics Could Reshape Diet Drug Capital and Regulation
The development of oral glucagon-like peptide-1 (GLP-1) receptor agonist therapies, moving from injectable to widely accessible pills, signals a profound but underappreciated shift poised to disrupt diet drug markets, regulatory regimes, and healthcare delivery models over the next decade. This insight paper unpacks a non-obvious inflection emerging from recent FDA approvals and Medicare policy adaptation that could recalibrate capital flows, industrial structures, and governance mechanisms beyond current hype around obesity drugs.
Starting with Novo Nordisk’s Ozempic oral tablet and Lilly’s soon-to-launch orforglipron (Foundayo), GLP-1 therapeutics in pill form combine efficacy with consumer convenience, triggering shifts in demand profiles, distribution networks, and payer strategies. The full impact of oral GLP-1 drugs will likely emerge as these medications expand access beyond specialty clinics towards broad population coverage, amplified by evolving reimbursement policies, notably Medicare’s planned weight-loss drug coverage commencing 2026.
Signal Identification
This phenomenon qualifies as an emerging inflection indicator manifesting within a medium to high plausibility band on a 5–10 year horizon. It extends beyond a mere pharmaceutical innovation to a systemic recalibration of chronic condition treatment paradigms that strategically exposes healthcare, pharmaceutical manufacturing, retail pharmacy, and health insurance sectors. It merits strategic intelligence focus because oral GLP-1 drugs may catalyse new regulatory approaches to drug classification, alter therapeutic adherence patterns, and reshape competitive dynamics in weight management and diabetes care markets.
What Is Changing
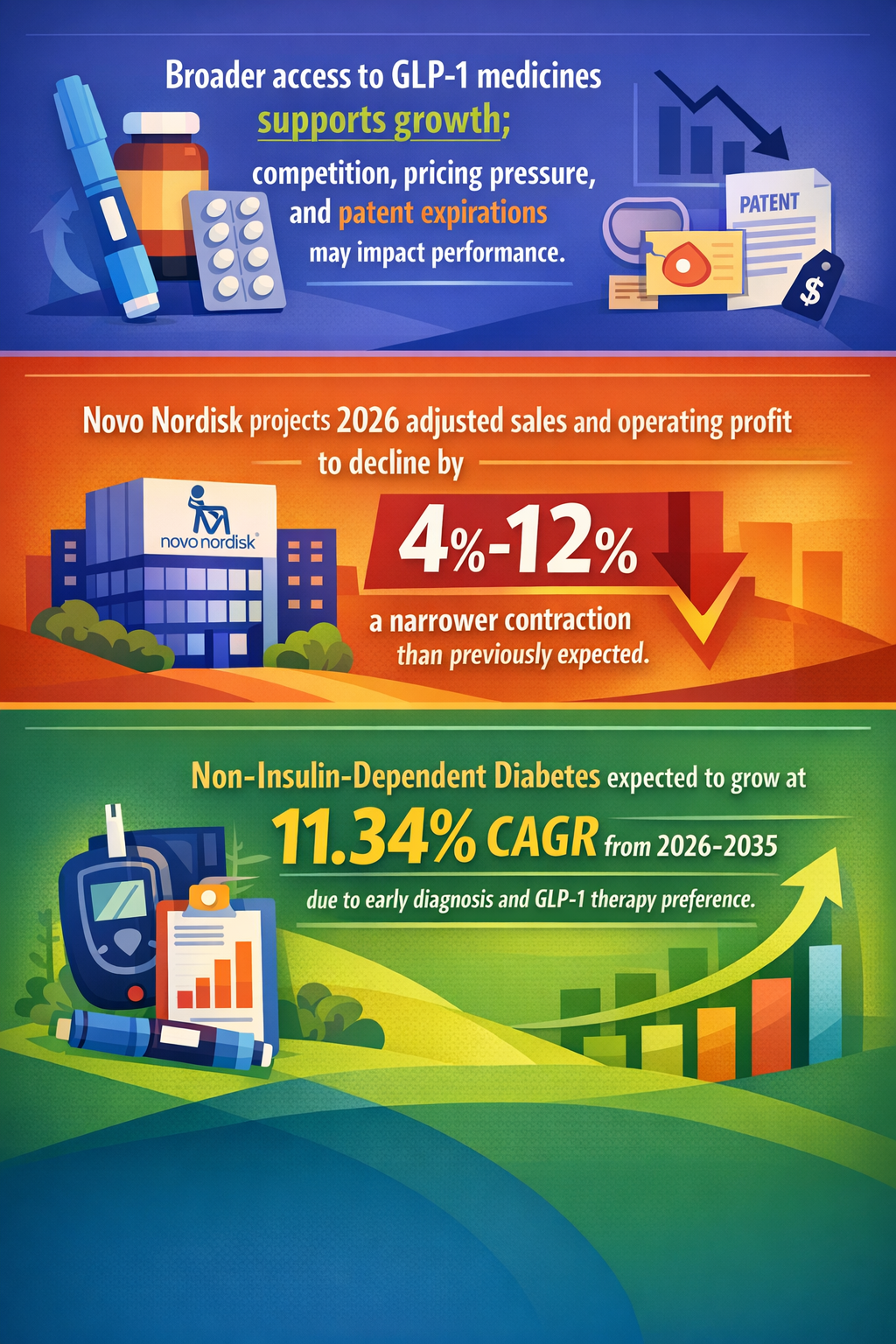
Recent approvals of oral GLP-1 drugs like Novo Nordisk’s Ozempic tablet, anticipated FDA decision by end of 2026, and Eli Lilly’s orforglipron pill, introduc e a market-first: oral peptide-based therapies effective for obesity and type 2 diabetes (Pharmaceutical Technology 25/04/2026; BioPharma Dive 15/04/2026). This marks a significant departure from previous reliance on costly, injection-only formats with limited patient adoption due to convenience barriers.
Further amplifying this shift, Medicare’s unprecedented decision to begin covering GLP-1 weight-loss drugs starting 2026 represents a watershed in pharmaceutical reimbursement policy, signalling broad public payer interest in addressing obesity as a chronic, treatable condition (KFF Health News 22/04/2026; PR Newswire 01/07/2026).
Together, these developments integrate into a systemic theme of “oralization” of peptide therapeutics—a novel concept with large-scale implications. This evolution enhances patient adherence, expands distribution through retail pharmacies nationwide (over 70,000 outlets for Ozempic), and fosters a consumer-centric paradigm shift where chronic disease management can be more decentralized and digitized (PR Newswire 25/04/2026).
Additionally, peak sales for GLP-1 therapies are forecasted for 2026, reflecting a potential industry plateau but also an inflection where innovation shifts from injection-based therapies to second-generation oral formats with enhanced convenience and consumer appeal (Business20Channel 05/05/2026). The pipeline competitiveness intensifies with trillions of dollars expected to flow into obesity and diabetes management drug markets estimated to reach $150 billion by 2030 (The National News 25/04/2026).
Disruption Pathway
The rise of oral GLP-1 drugs will likely accelerate if regulatory agencies adopt expedited pathways and standardize oral peptide classifications, moving away from treating peptides strictly as injectables or biologics with complex manufacturing oversight. This could lower barriers for market entry and widen therapeutic options beyond incumbent manufacturers.
As patient preference increasingly favours oral forms for ease of use and reduced stigma, adherence and completion rates may rise, enhancing long-term efficacy assessments and outcomes-based reimbursement models. Widespread adoption stresses current specialty pharmacy-centric distribution systems, compelling reconfiguration toward retail pharmacy involvement and direct-to-consumer digital health integrations.
These shifts may intensify competition, forcing dominant players to innovate delivery models, pricing structures, and subscription or adherence support services—as seen in Novo Nordisk’s subscription programs for Wegovy (MMM-Online 23/04/2026).
On the regulatory front, expanded Medicare coverage catapults GLP-1 drugs from niche prescription markets to broad public health interventions, possibly triggering new cost-effectiveness frameworks and payer-provider negotiations that recalibrate risk and liability assumptions around obesity treatment.
Unintended consequences may include overprescription risks, heightened supply chain pressures, and politicization of obesity treatment funding, which could invite stricter governance or tighter access controls. Feedback loops in patient outcomes, regulatory adjustments, and capital investment cycles will redefine industrial strategies through the 2030s.
Why This Matters
For capital allocators, the shift to oral GLP-1 therapeutics suggests a reshuffling of pharmaceutical R&D priorities and supply chain investments towards oral peptide formulation technologies and manufacturing infrastructure in the U.S., as exemplified by Ozempic’s fully domestic production (PR Newswire 25/04/2026).
Regulatory bodies may face pressures to finalize peptide classification frameworks that reduce ambiguities between biologics, small molecules, and novel modalities—a shift with ripple effects in drug approval, manufacturing oversight, and pharmacovigilance standards. Health insurers and public payers will need to develop analytics and policy instruments to manage expanded coverage responsibly without unintended cost overruns.
Industrial incumbents and emerging innovators will need to strategically position themselves in oral peptide delivery, digital adherence monitoring, and subscription access models to capture the next wave of market share.
Failure to anticipate these shifts may expose stakeholders to liability risks, supply shortages, and reduced competitiveness.
Implications
This development may structurally transform the diet drug and chronic disease treatment markets by normalizing oral peptide therapeutics, shifting chronic condition management from specialist clinics to primary and retail care settings, and embedding medication adherence within digital ecosystems. This could erode traditional biologics’ injection-based business models and reorient capital flows towards oral formulation platforms.
This signal is unlikely to represent a temporary "fad" driven by media attention to weight loss drugs but instead could indicate a durable industry evolution given regulatory and payer institutionalization. However, alternative interpretations might see oral GLP-1 drugs as expensive stopgaps constrained by long-term safety or efficacy limitations or by emerging competitors from gene therapies and microbiome interventions.
Early Indicators to Monitor
- Regulatory classification decisions and issuance of oral peptide-specific guidance documents.
- Volume and geographic spread of Medicare and private payer formularies including oral GLP-1 drugs.
- Capital investment surges in oral peptide manufacturing scale-ups and formulation tech startups.
- Pharmacy procurement pattern shifts reflecting mass adoption outside specialty channels.
- Registries and real-world data demonstrating adherence improvements unique to oral therapies.
Disconfirming Signals
- Regulatory delays or rejection of oral GLP-1 applications undermining credibility of oral peptides.
- Reversal or scaling back of Medicare GLP-1 coverage due to budget constraints or safety concerns.
- Supply chain disruptions causing shortages leading to patient and clinician distrust.
- Emergence of serious adverse events undermining oral GLP-1 benefit-risk profiles.
Strategic Questions
- How will shifting regulatory classification of oral peptides impact competitive dynamics and market entry barriers?
- What adjustments must payers and providers plan for to manage expanded GLP-1 access without compromising sustainability or equity?
Keywords
oral GLP-1 therapeutics; peptide drug delivery; Medicare coverage; regulatory frameworks; chronic disease management; pharmaceutical manufacturing; healthcare innovation
Bibliography
- Non-Insulin-Dependent Diabetes is expected to register the fastest CAGR of 11.34% during 2026-2035, driven by early diagnosis and increasing preference for GLP-1 therapies. Yahoo Finance. Published 22/04/2026.
- Plus, for some patients, Medicare will start covering GLP-1 drugs for weight loss in 2026. KFF Health News. Published 22/04/2026.
- Next week's launch of Foundayo, known scientifically as orforglipron, will come four months after Novo's Wegovy pill. BioPharma Dive. Published 15/04/2026.
- Medicare GLP-1 Bridge will be available nationwide across all states and territories, providing eligible patients access to all doses and formulations of Wegovy injection and Wegovy pill, and will run through the end of 2027. PR Newswire. Published 01/07/2026.
- Novo Nordisk to launch Ozempic, the only FDA-approved oral peptide GLP-1 medication for adults with type 2 diabetes, available through 70,000+ US pharmacies nationwide. PR Newswire. Published 25/04/2026.